Matter Waves :
In 1924, Louis de Broglie first proposed the existence of matter waves.
He supported the wave-particle dual nature of radiation and proposed the existence of matter waves. In his hypothesis, he suggested that the wave aspect of the matter is related to its particle nature in the same quantitative way that is in case of radiation.
The total energy 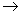 related to the frequency related to the frequency 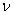 of the wave. of the wave.
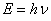
and the momentum p is related to the wavelength 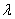 of the wave. of the wave. |