| |
| In 1909: |
| R. A. Millikan developed the famous “Oil-Drop Experiment” to determine the charge of the electrons from the charge of the oil droplets and found that the charge is integrated multiple of electrical charge.
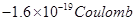 the charge of electron. the charge of electron.Also, with the help of this experiment, the mass of the electron was calculated as
|
| In 1909: |
| Ernest Rutherford (England) demonstrated that the atom is mostly empty space with a small positively charged nucleus containing most of the mass and low mass negatively charged particles (Thompson’s electrons) orbiting this nucleus. Rutherford could experimentally identify nuclear particles with positive charge that he called protons. Although he could explain the charge of atomic nuclei with the right number of protons, the mass of the nucleus for large atoms was always larger than the sum of its protons. Therefore he postulated the existence of a neutral particle with a mass nearly the same as the proton which, when added to the protons in the nucleus, would give the right mass. Rutherford called this hypothetical particle the neutron. Later (1930) Rutherford’s colleague James Chadwick was able to detect the neutron experimentally. |
|
|
|