| |
| Conclusions: |
- Since most
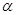 particles passed through the atoms without being deflected - An atom must be having mostly empty space. particles passed through the atoms without being deflected - An atom must be having mostly empty space.
- The
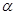 particles occasionally strike a small strongly scattering region. This region could be a concentrated positive charge. particles occasionally strike a small strongly scattering region. This region could be a concentrated positive charge.
- Since atoms have no overall electrical charge (atoms are neutral), there must be just enough negatively-charged particles outside the nucleus to just balance the positively-charged nucleus.
Therefore,
- The Rutherford's experiment suggested that the atom consists of mostly empty space with a very small positively-charged nucleus, outside which are just enough negative charges to equal the positive charge in the nucleus.
- Since the electrons do not crash into the nucleus but exist in stable atoms, this suggests a planetary model with negatively charged electrons circulating around a positive core.
|
|
|
|