Converting reaction for the entire blow:
Slag formation stage :  and and |
(3) |
Blister formation stage:  |
(4) |
From the amounts of 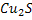 and FeS oxidised, one can calculate the amount of oxygen and hence amount of air for both the stages. and FeS oxidised, one can calculate the amount of oxygen and hence amount of air for both the stages.
The amount of air 24325.3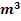 . .
Time required for slagging stage = 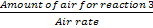
Time required for blister copper formation stage = 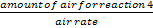
On substituting the values we get time required for slagging stage = 184 minutes and time required for blister copper formation stage = 86 minutes
From reactions 3 and 4 we get
% 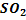 in slag formation = 15% and in blister copper stage = 21% in slag formation = 15% and in blister copper stage = 21%
Let X kg FeS remains in matte after oxidation. From the definition of the matte grade
0.6 = 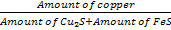
Moles of FeS remaining = 44.1. Moles of FeS to be oxidised = 59.4
Air required = 9504 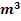
Heat generated/mimute can be calculated from reaction 3 and 4 in that we use the moles of reactants participating in the reactions. The following values may be used:
Heat of formation of FeO at 298K = -64300 kcal/kg.mole
Heat of formation of FeS at 298K = -23100 kcal/kg.mole
Heat of formation of 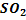 at 298K = -70940 kcal/kg.mole at 298K = -70940 kcal/kg.mole
Heat generated in slag formation stage = 63 x 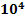 kcal/minute kcal/minute
Heat generated in blister copper stage = 4.4 x 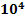 kcal/minute kcal/minute
|