Composition of a mixture
It is expressed in terms of mole fraction or mass fraction. If mixture conatins n, moles of component 1,n2 moles of component 2, n3 moles of component of 3.; then mole fraction of ith component is
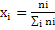
Similarly mass fraction of ith component in the mixture is given by.
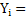 
Derivation of unit of universal constant (R)
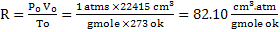
In CGS unit
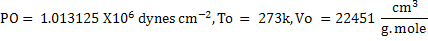
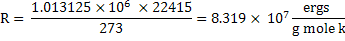
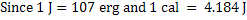
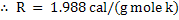 . .
In SI units.
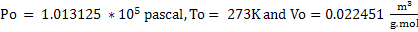
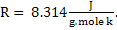
Similarly in M K S system 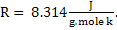
Current =Ampere (A). In solids current consists of electron flow. In electrolyte solutios.most of the current flow by motion of ionic species for example 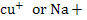
1coulomb is unit of charge: flow of 1A/s.
SI unit of electrical potential is volt. Volt is the potential in which the charge of 1 coulomb experiences a force of 1 Newtron.
SI unit of resistance is ohm. Ohm is defined as the resistance which permits flow of 1 A current under an imposed electrical potential difference of 1V.
Some basic equations of electrical flow are:
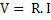
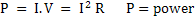
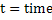
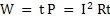 . W=energy measured in joules and power in watts . W=energy measured in joules and power in watts
A Faraday is one mole of electrons.
1 faraday = 96500 coulomb. One faraday will discharge one gram equivalent of ions.
The liberation of one g equivalent of any metal consumes 96500 coulombs of electricity
How many gram moles of 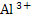 ions could be discharged in one minute by ions could be discharged in one minute by 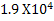 A current, if no loss of current occur. A current, if no loss of current occur.
In one minute a current of 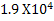 A will carry A will carry 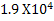 coulombs of electricity. coulombs of electricity.
Gram moles of Al deposited 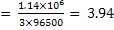
|