| |
| 2 Formation and evolution of microstructures |
| 2.1 Motivation |
Shown below is the Pb-Sn phase diagram (Figure 1). Consider the three alloys of the given compositions marked in the phase diagram, all of which are cooled from 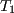 to to 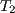 . How different would the three microstructures be? . How different would the three microstructures be? |
| 2.2 Phase diagrams and microstructures |
Let us consider the Ph-Sn phase diagram shown in Figure 1. There are three compositions that are considered. These three compositions correspond to eutectic, hypo-eutectic and hyper-eutectic systems. In the case of eutectic system, the microstructure is as shown in Figure 2: it consists of a fine mixture of the two phases, namely, 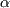 and and 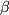 , the tin-rich and tin-poor phases. In the case of hypo- and hyper-eutetic systems, on the other hand, the microstructures are as shown Figure 3. These type of microstructures arise, as shown schemicatally in Figure 4, due to the formation of the pro-eutectic , the tin-rich and tin-poor phases. In the case of hypo- and hyper-eutetic systems, on the other hand, the microstructures are as shown Figure 3. These type of microstructures arise, as shown schemicatally in Figure 4, due to the formation of the pro-eutectic 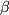 (or (or 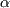 )phase first making the composition of the remaining liquid shift towards the eutectic composition; the liquid that remains on achieving the eutectic point in terms of composition and temperature, then, phase separates into the eutectic mixture. Thus, from this exercise, it is very clear that the phase diagrams give us some indication of the microstructures that can result. However, phase diagrams assume equilibrium and hence gives an idea of microstructure that can form due to very slow cooling (small values of cooling rate). If we want to understand the formation of microstructures, taking into acocunt the cooling rates, we need to peruse TTT or CCT diagrams. )phase first making the composition of the remaining liquid shift towards the eutectic composition; the liquid that remains on achieving the eutectic point in terms of composition and temperature, then, phase separates into the eutectic mixture. Thus, from this exercise, it is very clear that the phase diagrams give us some indication of the microstructures that can result. However, phase diagrams assume equilibrium and hence gives an idea of microstructure that can form due to very slow cooling (small values of cooling rate). If we want to understand the formation of microstructures, taking into acocunt the cooling rates, we need to peruse TTT or CCT diagrams. |
|
|
|