In this section, we develop a simple bond-breaking model to calculate the surface energy of a solid; we assume that the solid is in contact with its own vapour; we also assume that the temperatures are low enough that the primary contribution to the surface energy comes from the broken bonds.
Consider a simple cubic lattice with a lattice parameter of 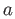 . Let us consider a surface that makes an angle of . Let us consider a surface that makes an angle of 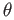 with one of the principal axes of the crystal. The crystalline surface (of unit length) is shown schematically in Figure 7. The open circle marked 1 represents an atom in the bulk; it has formed four bonds in the plane of the figure; if we consider the crystal to be infinite in the direction perpendicular to the plane of the figure, it has also formed two bonds in the perpendicular direction: one above the plane and another below. The filled circle, which represents a surface atom has formed only two bonds out of the four that it can form in the plane of the figure. These dangling bonds are the ones that contribute to the enthalpy (in this case, internal energy) of the interfacial free energy. with one of the principal axes of the crystal. The crystalline surface (of unit length) is shown schematically in Figure 7. The open circle marked 1 represents an atom in the bulk; it has formed four bonds in the plane of the figure; if we consider the crystal to be infinite in the direction perpendicular to the plane of the figure, it has also formed two bonds in the perpendicular direction: one above the plane and another below. The filled circle, which represents a surface atom has formed only two bonds out of the four that it can form in the plane of the figure. These dangling bonds are the ones that contribute to the enthalpy (in this case, internal energy) of the interfacial free energy. |