How do Atoms assemble into solid structure?
From structural point of view, there are three ways in which atoms can pack together.
Accordingly, the materials are grouped as
- Crystalline,
- Amorphous and
- Semi-crystalline
In Crystalline structures, the atoms pack in a regular periodic form which
is typical of metals, ceramics and for a very few polymers. A typical crystalline structure is shown below. Crystalline
structures are often grouped into three categories: Single Crystal, Poly-
Crystal and Liquid Crystals. Single crystal as the name suggests consists
of only one large crystal of the material. Though it is very expensive to
develop, single crystals being free from most of the defects show unusually
high mechanical and other functional properties. Hence, in many
specialised applications they are in very high demand. For example,
Titanium based super alloys are mostly developed as single crystals to use
in Turbine blades.
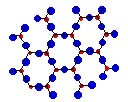
Crystalline Structure
Exactly opposite to the crystalline structures, the amorphous structure
refers to completely irregular packing of atoms. Many polymers show this
kind of packing. Also, rapid cooling of some of the materials from the liquid
phase result in amorphous or glassy form of structure. A typical solid in
amorphous form is shown below:
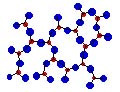
Amorphous Structure
Semi-crystalline structures are placed in between the two extremes.
Some times glassy structures show local regularities. The degree of
regularity determines the degree of crystallinity of such materials. Polymers like low density polyethylene (LDPE), Polystyrene etc. fall into this catagory.
|