Step Polymerization
In a step reaction mechanism, monomers disappear very early since they
can react with any nearby monomer. In contrast to chain polymerization, no
special activation is needed to allow a monomer to react. Consequently, the
molecular weight of the polymer rises all through-out the reaction.
Frequently these reactions are copolymerizations, where two types of
monomers are present and each reacts only with the other (and not with
monomers like itself).
This type of polymerization is also called condensation polymerization
because often water come out as a by-product of polymerization.
Some of the examples of Step Polymerization are:
•Nylon
•Polyurethane
•PET
CURING of Thermoset Polymers
Thermoset Polymers like epoxy resin, often needs to be cured for some
hours before they could be put into applications. The curing actually refers
to the growth of networking chains in the polymer which is accelerated by
the application of heat, pressure and hardeners.
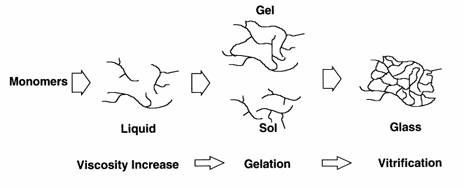
Viscosity of a polymeric system during curing rises until Gelation occurs.
At this point, two phases exist: a gel phase and a sol phase. The gel phase
is the gelled part; whereas the sol phase can be extracted with solvents.
The amount of sol phase present decreases as the reaction progresses
further. Upon further reaction, vitrification (hardening) occurs.
|