Types of Solid Solution
There are two types of solid solution in metals: Substitutional and
Interstitial
Substitutional – In this form of alloying, solute atoms replace or
substitute the host atoms e.g. Au-Ag. The following parameters
are important for substitutional solutions:
Difference between atomic radii of the solute and the solvent
atoms should be within ± 15 %. The other important issues are -
- Electronegativity
- Relative valencies of participating atoms
- Crystal structures of metals of both atom types should be the
same.
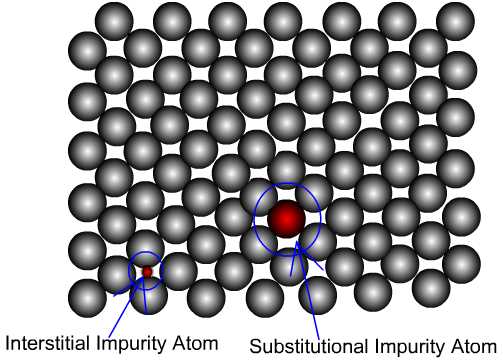
Interstitial – When the difference in radii between the participating
atoms is too high, the alloying could occur in the form of interstitial
filling. Impurity atoms fill voids or interstices among host atoms
e.g. Fe-C. The figure below shows substitutional and interstitial
alloying schematically.
|