2.12. Assignments
- Write down the structure of 22nd natural amino acid, its 3 letter code and single letter code.
- Write down any one structure, name, 3-letter code and one letter code of amino acid of each category: (a) Neutral amino acid with hydrophilic side chain, (b) Acidic Amino Acid and (c) basic amino acid.
- Name the main three types of protein’s secondary structures. Write down the general structures of β3- and β2-amino acids.
- What do you mean by genetic alphabet, genetic codon and genetic code?
- Define the followings: (a) Chemical Biology, (b) Biochemistry, (c) Bio-organic Chemistry, (d) Proteomics and (e) Glycobiology
- Answer the followings:
- If three different amino acids (gly, ala, leu) are used to make a tripeptide, how many different sequences are possible?
- 6
- 9
- 3
- 12
- The primary structures of methionine-enkephaline is: tyr - gly - gly - phe – met. The N-terminal and the C-terminal amino acids respectively are:-
- met- and gly-
- Tyr- and gly -
- tyr – and phe –
- tyr - and met-
- Normal hemoglobin is apparently more polar and soluble in water because of the presence of polar amino acid glutamic acid but Sickle Cell hemoglobin is more non-polar and insoluble. The non-polar amino acid present in Sickle Cell hemoglobin is-
- Isoleucine
- Leucine
- Valine
- Alanine
- The order of increasing polarity of the amino acids-Ser; Glu; Asp; Lys; Ala; Gln is
- Ser > Glu > Asp > Lys > Ala > Gln
- Ser > Glu > Asp > Lys > Gln > Ala
- Glu > Ser > Asp > Lys > Gln > Ala
- Ala < Lys < Ser < Glu < Asp < Gln
- If three different amino acids (gly, ala, leu) are used to make a tripeptide, how many different sequences are possible?
- Explain the differences between primary, secondary, tertiary, and quaternary protein structures by giving brief definitions of each. What types of bonding are used in each?
- Explain the difference between the alpha helix and the beta pleated sheet protein structures. What are the differences in the hydrogen bonding?
- Write down the structure of A, B, C and the absolute configuration of the produced chiral aminoacid in the following scheme.
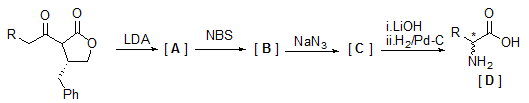
- Why the solid phase peptide synthesis proceeds from C to N-terminus and not N to C terminus synthesis?
- What are the main applications of unnatural amino acids?