|
Sodium naphthalide is an example of an organometallic salt with a delocalized radical anion, C10H8-.
Such compounds are readily prepared by reacting an aromatic compound with an alkali metal in a polar aprotic solvent.
Naphthalene dissolved in THF reacts with Na metal to produce a dark green solution of sodium naphthalide. 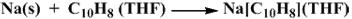
EPR spectra show that the odd electron is delocalized in an antibonding orbital of C10H8.
Formation of radical anion is more favorable when the π of LUMO of the arene is low in energy.
Simple MOT predicts that the energy of LUMO decreases steadily on going from benzene to more extensively conjugated hydrocarbons.
Sodium naphthalide and similar compounds are highly reactive reducing agents.
They are preferred to sodium because unlike sodium, they are readily soluble in ethers. |