ClO
2- Total number of electrons 7 + 2 x 6 + 1 = 20 = 10 pairs
Identify the central atom and connect the peripheral atoms with a pair of electrons as two dots and count the remaining electron pairs
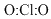
20-4 = 16 electrons left (8 pairs)
Complete the octet of oxygen atoms and count the remaining electron pairs
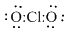
16-12 = 4 electrons
Complete the octet of chlorine atom,
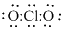
Since all the electrons are utilized and octet is satisfied, no need of any multiple bonds.
The structure is
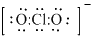
or
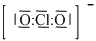
or
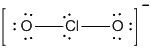
CO (carbond monoxide) Total number of electrons 4 + 6 = 10
Connect the two atoms with a pair of electrons as two dots and count the remaining electron pairs
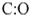
10-2 = 8 electrons left
Complete the octet of oxygen atoms and count the remaining electron pairs
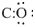
8-6 = 2 electrons
Place the remaining electron pair on carbon atom,
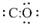
All the electrons are utilized and octet is not satisfied for carbon as there is a shortage of four more electrons.
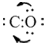
Drag two electron pairs on oxygen atom in between carbon and oxygen to establish two more bonds so that a triple bond exists between C and O which satisfies the octet of both C and O.
The structure is
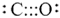
or

or
