| |
The size of the simulation system is so chosen such that the number density in the simulation box corresponds to the experimental density of the system. Argon is a liquid at 85 K with density 1.40 gm/cc. Corresponding to this density at this temperature, 64 argon atoms are to be kept in a volume of 3.03×103Å3. (See problem 1 at the end of this chapter. Convert the density from gm/cc to number of particles per Å3by converting grams into moles and multiplying by Avogadro number. Also use, 1 cc = 1024Å3)The simulation box is usually taken to be cubic, this makes the box size to be 14.4744 A. |
In order to prepare an initial configuration for the system, one may start with a simple cube, in which the argon atoms are placed on to the lattice sites. Given these initial positions r(t = 0) to the atoms, we need to choose an appropriate time step, Δt. |
The potential energy function for argon is a simple Lennard-Jones 12-6 potential, |
|
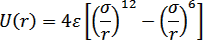 |
(27.1) |
|
Figure 27.1: The Lennard-Jones potential defined by the two parameters, s (particle diameter) and e (well-depth). The deeper the well-depth e, the stronger is the interaction between the two particles. |
|
|
|