Slow Coagulation
As shown in figure 10.17, there is some energy is required for coagulation to occur. Due to this energy barrier the coagulation is slow. Energy barrier arises due to repulsive component in the force. As done for rapid coagulation, number of particles crossing a shell of radius ‘r’ towards a reference per second is
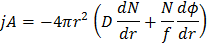
First term is trying to coagulate while the second term is driving the flux in opposite direction. So it makes the kinetic of coagulation slow. Removing the restriction that the reference is stationary (replacing D with 2D) we get:
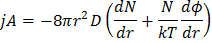
Or,
which is a first order homogenous equation. For steady state, flux (jA) is constant so this differential equation can be solved with appropriate boundary conditions. As 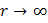 , , 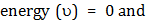 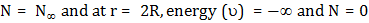 . So equation (10.25) has solution as . So equation (10.25) has solution as
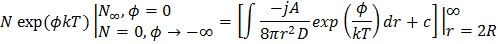
Above equation can be written as
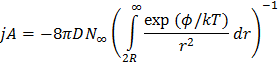
|