Equating these 2 values of σ we can solve for ψo as a function of [H+]B .
Let us look at some typical values: 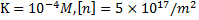
Table 2: Effect of H+ ion concentration on zeta-potential
[H+]B |
ψo(mV) |
10-4 |
-163 |
10-3 |
-91 |
10-2 |
-31 |
So we see that |ψo| decreases when [H+]B increases.
What will be the effect if more salt is added?
In that case, value of κ will not be affected by [H+]B. Then the above done derivation does not hold.
|