Debye-Huckel Approximation when potential on the surfaces are not same
Now we consider a more general case when potential on the surfaces are not same. For this case we take Debye-Huckel approximation to be valid that is taking surface potential to be low. For this Poisson Boltzman equation becomes
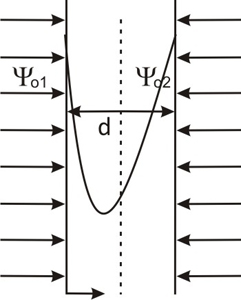
Fig. 9.4: Distribution of potential between two charged particles.
General solution of above equation is
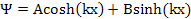 |
(9.38) |
By applying the appropriate boundary conditions we can determine the constants in the above equation. Let us assume that the potential at two surfaces are ψo1and ψo2 as shown in the figure. Thus the boundary conditions we have are
1) ψ = ψo1 at x= 0;
2) ψ = ψo2 at x= d
Substituting these boundary conditions in equation (9.38) we have
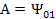
and
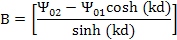
Thus
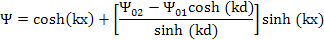 |
(9.39) |
Since the potential at surfaces are different so the minimum of potential (ψm) will lie somewhere in between 2 plates but not at the centre. If we take ψo1 = ψo2 = ψo then we get ψm at the middle point as:
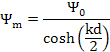
|