It may be noted that eqn. 7.92 is the same as 6.162 (section 6.15), which describes the Lewis -Randall rule. Thus when Henry’s law is applicable for the solute then Lewis-Randall rule is applicable for the solvent. Since for a system temperature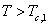 the fugacity the fugacity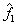 of pure liquid phase for ‘1’ is hypothetical, it follows that the Henry’s law constant of pure liquid phase for ‘1’ is hypothetical, it follows that the Henry’s law constant 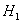 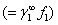 is necessarily a hypothetical quantity as well. Since solubility of a gas is temperature dependent, it follows that is necessarily a hypothetical quantity as well. Since solubility of a gas is temperature dependent, it follows that 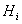 is also a function of temperature. The Henry’s law constant for a large number of gases with water as the solvent has been reported in the literature. For example for acetylene the value is 1350bar, for carbon dioxide 1670bar, and for air 72950bar). Fig. 7.14 presents the value of Henry’s law constant for a number of gases in water as a function of temperature. is also a function of temperature. The Henry’s law constant for a large number of gases with water as the solvent has been reported in the literature. For example for acetylene the value is 1350bar, for carbon dioxide 1670bar, and for air 72950bar). Fig. 7.14 presents the value of Henry’s law constant for a number of gases in water as a function of temperature. |