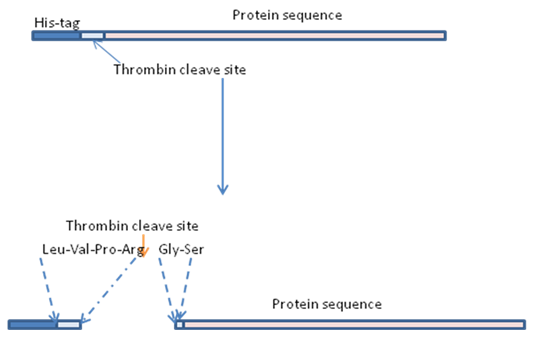
Figure 2: His - tagged Proteins with Thrombin cleavage site . First, his-tagged protein is purified by Immobilized-metal affinity chromatography . After purification, t ag is removed by thrombin cleavage. After thrombin cleavage, the sample is again passed through Immobilized-metal affinity chromatography column . The cleaved protein will come in elution while the tag will remain bound to column.
Immobilized-metal affinity chromatography (IMAC) was first used to purify proteins in1975 by Porath and group using the chelating ligand iminodiacetic acid (IDA, Figure). IDA was charged with metal ions such as Ni 2+ , and then used to purify a variety of different proteins and peptides. IDA has only 3 metal-chelating sites and cannot tightly bind metal ions. Weak binding leads to ion leaching upon loading with strongly chelating proteins and peptides or during wash steps. This results in low yields, and metal-ion contamination of isolated proteins
(Fig.3 and Fig. 4).
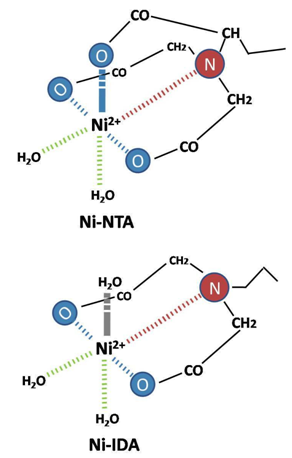
Figure 3: Comparison of interaction of different metal chelate matrices with nickel ions. |