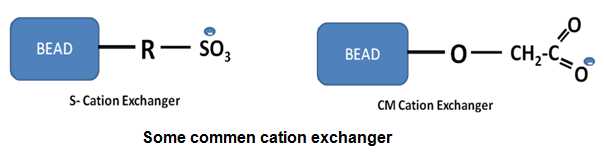
Cation Exchange Chromatography: Commonly used cation exchange resins functional groups are following:
| Carboxymethyl (CM) |
-O-CH2-COO - |
Sulphopropyl (SP) |
-O-CH2-CH2-CH2-SO3- |
Methyl sulphonate (S) |
-O-CH2SO3- |
Solid support with these functional groups can be prepared with various beads. They differ in few properties like flow rate, stability, binding capacity (linked with porocity) etc. Cation exchangers based on dextran (Sephadex), agarose (Sepharose) and cross-linked cellulose (Sephacel) were the ion exchange matrices with high porosity, leading to improved flow properties and high capacities for macromolecules. Typically, cation exchange chromatography is performed using buffers at pH's between 4 and 7 and running a gradient from a solution containing just this buffer to a solution containing this buffer with 1M NaCl.
The surface charge on the molecules (proteins, nucleic acids etc), which binds to cation exchanger should be net positive. Thus, to get binding of a specific protein pH should be below the pI of that protein. Once salt concentration increases, it results in increase in Na + (cation) and Cl - (anion). Beyond a certain point, positively charged protein is exchanged with cation. Thus, this type of chromatography is called cation exchange chromatography (Fig. 1 and Fig.2).
|