One very simple example of rational protein design: Designing an inactive form of Pancreatic ribonuclease A
Pancreatic ribonuclease A is an enzyme comprising of 124 amino acids that cleaves the covalent bonds that join ribonucleic acids (RNA). From available literature about the protein we know that histidine at position 119 is necessary for catalysis. Thus, if at position 119 in the sequence the naturally occurring histidine is replaced with an alanine, the mutant protein is referred to as a histidine 119 ? alanine (H119A) mutant of ribonuclease A. This mutant protein is expected to have little or no biological activity, because histidine 119 is important for catalytic activity. A simple example of mutation is shown in Fig. 2.
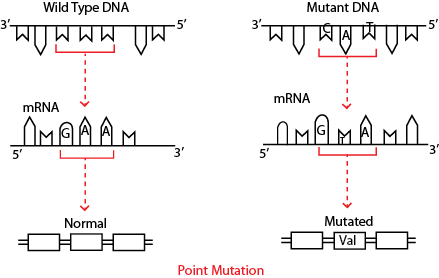
Figure 2: Diagrammatic representation of a designed mutation in protein at DNA level.
Another example of rational protein design: A very common example of Protein Engineering is engineering of enzyme subtilisin, a protease (a protein-digesting enzyme). To improve the efficiency of laundry detergents, subtilisin is added in the detergent. However, subtilisin is inactivated by bleach. Experimental and structural analysis revealed that this inactivation was due to oxidation of the amino acid methionine at position 22 of the subtilisin molecule. Using site-directed mutagenesis technique the subtilisin gene in E. coli was mutated and methionine was changed by alanine. This engineered subtilisin showed high activity and stability, and now many laundry detergents contain cloned, engineered subtilisin.
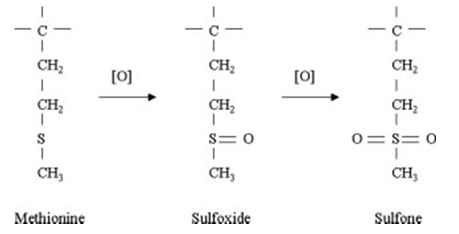
Methionine is immediately adjacent to a catalytic serine residue and therefore is located at a particularly sensitive structural position. Methionine oxidation results in increased side chain bulk or perhaps even the introduction of strongly electronegative oxygen atom(s) in the immediate vicinity of the active site negatively effects the catalytic activity.
|